A dc protein assay is used to quantify the amount of total protein in a sample. There are two main ways to do this, and both involve a color change as an indicator of protein presence, which means they are colorimetric assays:

ELISA stands for Enzyme Linked Immunosorbent Assay. The immunoassay utilizes the specific lock-and-key recognition between antibodies and antigens. This recognition occurs naturally in the adaptive immune system; antibodies are created by the immune system when an antigen such as a virus or bacteria invades the body. The immune system recognizes the foreign invader and creates antibodies that specifically recognize surface proteins on the virus or bacteria. The antibodies are either attached to the surface of an immune cell or move freely through the body to tag the invader and begin a cascade of destruction and elimination. The most useful part of this process from a biotechnology and engineering perspective is the specificity of the antibody-antigen recognition.

Fluorescent microspheres are small polymers embedded with fluorescent dye. They are a useful tool for medical imaging because they are non-toxic and non-biologically reactive when used as directed. Fluorescent microspheres are also useful in research laboratories as markers for fluorescent microscopy and as standards for flow cytometry fluorescent cell sorting. The main benefit of using a polymer microsphere embedded with fluorescent dye rather than using the dye alone, is two-fold: the matrix protects the dye from photobleaching, and the microsphere concentrates the dye leading to a more robust fluorescent signal.
The Enzyme Linked ImmunoSorbant Assay (ELISA) is the gold standard immunoassay for clinical diagnosis of disease. The basis of any immunoassay is the specific molecular recognition between antibody and antigen. This is something that the immune system does naturally. The production of monoclonal antibodies in a laboratory has become commonplace and standardized, which makes it possible to use monoclonal antibodies in immunoassays such as an IgG ELISA. The antibodies are easy to purchase from commercial vendors, and they come with quality control reports ensuring that they will recognize the target antigen.

Protein purification services are available for anyone who is in need of a custom antibody or recombinant protein for research and development purposes. If your laboratory is not equipped to produce recombinant proteins in house, then this may be an attractive option. These services require you to provide a sequence and preferred expression system; in turn they will deliver a quality controlled protein with accompanying documentation to you within just a few weeks.

Molecular diagnostics entails the analysis of biomarkers to help diagnose, track the progression of, or determine risk factors and prognosis of disease. Biomarkers have been identified within the realm of genomics, epigenomics, transcriptomics, proetomics, metabolomics, and lipidomics:

The ELISA or Enzyme-Linked Immunosorbent Assay is the most established method of protein detection and quantification. The technique is commonly used in a wide variety of applications spanning from the basic research bench to clinical laboratories. The ELISA is a labeled assay, which means that it requires the use of a label to detect an antibody-antigen binding event. The label is commonly a fluorescent probe, chemiluminescent system, or enzymatic colorimetric reaction.

Molecularly imprinted polymers (MIP) are relatively new diagnostic and therapeutic tool. MIPs are highly specific three dimensional polymer imprints of molecules. The power of this tool may not be immediately obvious, but it is indeed a very useful technology. The power of MIPs lies in their specificity of binding to target molecules. Before the invention of MIPs, the only way to achieve this type of specificity was through antibody-antigen binding, surface receptor-ligand interactions, or protein affinities such as streptavidin and biotin. The problem with all of these systems is that they need to already exist. But what about a target that doesn't have a known molecule with a natural affinity? Herein enters molecularly imprinted polymers.

Faster and more efficient methods of pathogen detection are in high demand. The traditional methods involve collection of patient blood or swab samples for multi-day cultures. These methods are time-consuming and require full laboratories with skilled technicians and sterile equipment. As such, they are not ideal for low-income areas or for rapid pathogen detection. There is a need for rapid pathogen technology and point-of-care diagnostic tools. Ideally, these technologies will come with a built-in validation protocol. Magnetic nanoparticles and molecularly imprinted polymers are good candidates for improved pathogen detection systems. An additional benefit to using magnetic nanoparticles is that the separation process is easy to quantitatively measure with a validation protocol.

If you look closely at the product information for many commercial antibodies, you will see that they are protein A purified. Protein A is a surface protein that was originally found in the cell wall of staphyloccoccus aureus bacteria. On the surface of bacteria it serves as a defense against the host immune system and allows the bacteria to survive longer and be more virulent. Protein A binds the Fc portion of IgG antibodies.
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human use (ICH) was founded in 1990 as a way to standardize the introduction of new drug substances to the worldwide market. The council wrote and maintains guidelines for how new pharmaceutical products must be tested for stability and quality before they can be approved for worldwide distribution. The guidelines protect consumers and allow new therapeutic drugs to reach patients across international borders more quickly. The ICH has written guidelines for the stability testing of new drug substances and products. There is a general document known as Q1A(R2) that outlines the details of every stability test that a new drug substance must undergo before being registered. These tests examine how the drug degrades in high temperature or high humidity over time. It outlines methods for defining the mechanism of degradation for the new drug, and how to test proposed protective packaging for efficacy. A supplementary document (Q1B) contains additional details specifically about photostability testing.
Enzymes are the catalysts for biochemical reactions. As such, they speed up the transition from reactants to products without being consumed in the process. Multiple enzymes can be found in every cell, from bacteria up through to humans. We as humans have found ways to exploit enzymes to produce food products, fuel, pharmaceutical products, biotechnological tools, sensors, and much more. The potential uses for enzymes are seemingly limitless. The creation of solid support structures with immobilized enzymes has improved our ability to reuse enzymes in a controlled manner for a variety of applications. Immobilized enzymes can be reused multiple times before their efficacy is lost. This allows them to be an affordable part of industrial processes.
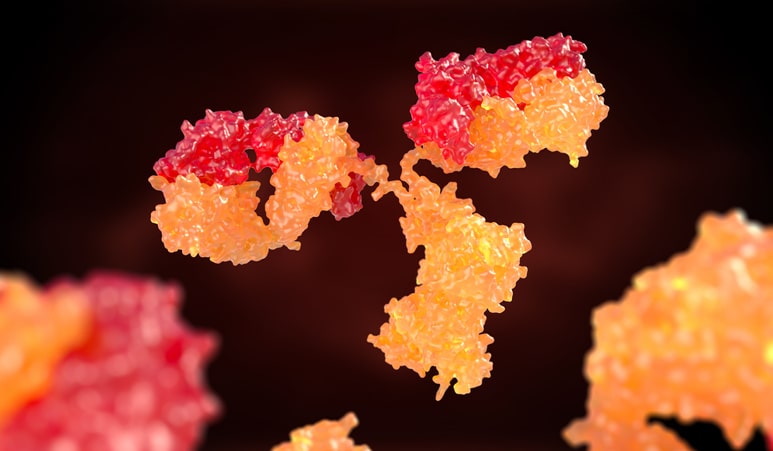
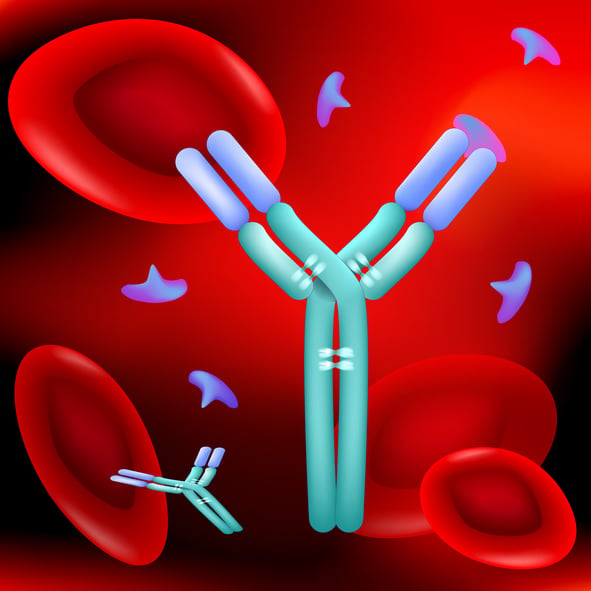
Until this point we have been thinking about antibodies as one of the five classes, IgG, IgE, IgD, IgA, or IgM. The basic unit of each antibody class is a Y structure, where the base of the Y is known as the Fc region and the arms are the Fab region. The entire IgG antibody is composed of four polypeptide chains (two heavy and two light). The Fc region is composed only of heavy chain, and the variable Fab region is built with heavy chain and light chain. The Fab region is where all of the antigen-binding occurs because the paratope, or antigen recognition site is located at the tip of each of the two arms of the Y. A single domain antibody paratope is made solely of a single heavy chain.
The structure of the most commonly known antibody class (IgG) was a mystery until 1959, when it was elucidated by Edelman and Porter . The duo approached the question from two very different directions, but they were both awarded the 1972 Nobel Prize in Physiology or Medicine for their groundbreaking work. Gerald Edelman said that he was, “fascinated by the specificity of antigen recognition by antibodies,” and hoped that, “by doing the primary structure of antibody molecules, the basis of their specificity would be revealed.” Indeed, it was.

Pharmaceutical validation is important to the manufacturing process to ensure product consistency and safety. It involves regulation of all raw materials and production procedures as well as testing of final product. The general rule of thumb is to follow good manufacturing practice (GMP). This demands that all protocols be up to date and followed by trained personnel. It also requires that equipment be well-maintained and inspected. In the case of clean-room usage the clean room needs to be verified.
We have come a long way from the days of blood letting, trephination, and snake oil salesmen peddling cure-all tonics. The oversight and regulation of organizations such as the European Medicines Agency and the Federal Drug Administration (FDA) have significantly improved the quality and safety of our medical and pharmaceutical products. Of course, our medical understanding has deepened dramatically, our science has become more sophisticated, and we have developed tools to perform large scale drug discovery and screening. With this deeper understanding of chemistry and drug development we have realized the importance of preserving the chemical molecules via proper storage conditions.
The ICH guidelines for stability lay out the requirements for identifying and maintaining drug efficacy by understanding the pathways of degradation. The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) was founded in 1990. The European Commission, FDA from the USA, and the Ministry of Health, Labour, and Welfare (MHLW), which later became the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan are all founding members. Since that time, many other regulatory authorities from around the world have joined the ICH. The stated mission of the ICH is to “achieve greater harmonisation worldwide to ensure that safe, effective, and high quality medicines are developed and registered in the most resource-efficient manner.”

In bacteria there are two main types of DNA—genomic and plasmid. Plasmid DNA is unique to bacteria. Eukaryotic cells don't typically have plasmid DNA unless it was put there by transfection for experimental purposes. The most important goal when isolating nucleic acids is to obtain the highest purity genetic material possible. When isolating genomic DNA it is important to remove plasmid DNA and RNA from the sample. Similarly, sometimes an experiment calls for the isolation of plasmid DNA, and the selective removal of genomic DNA is necessary. Also, some commercial RNA isolation kits include gDNA eliminator spin columns to remove genomic DNA from the isolate.
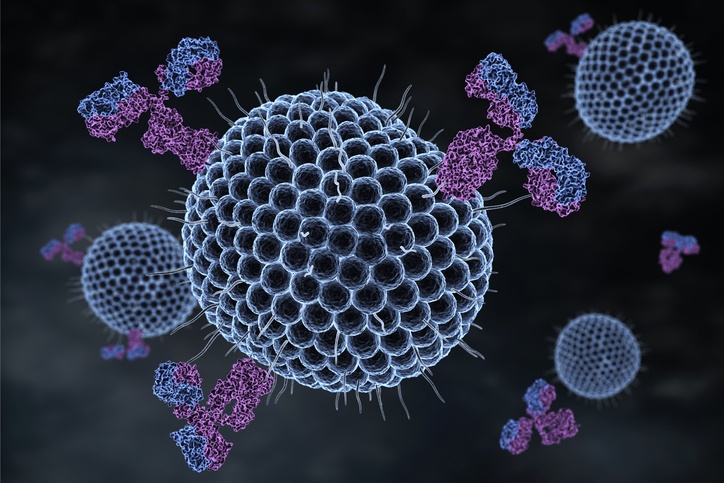
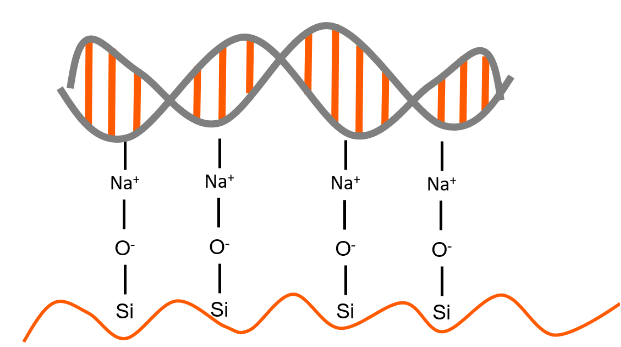
The concept of an antigen and antibody pair is central to modern medicine and biotechnology. These proteins match like a lock and key, with equisite specificity. The interactions are non-covalent, but have equilibrium constants ranging from 105 to 1012 M-1 Antibodies and antigens are proteins: polypeptide chains of amino acids. The IgG antibody is composed of four polypeptide chains, two heavy and two light, organized into a ‘Y’ shape. The base of the Y is called the Fc region, while the two tips are known as the Fab region.

After celebrating its IIVD seminars in Asia over the past few years MERCK has decided to organize this year’s event in Europe during the autumn season. This workshop will be held in Paris, on October 18th and 19th 2018.
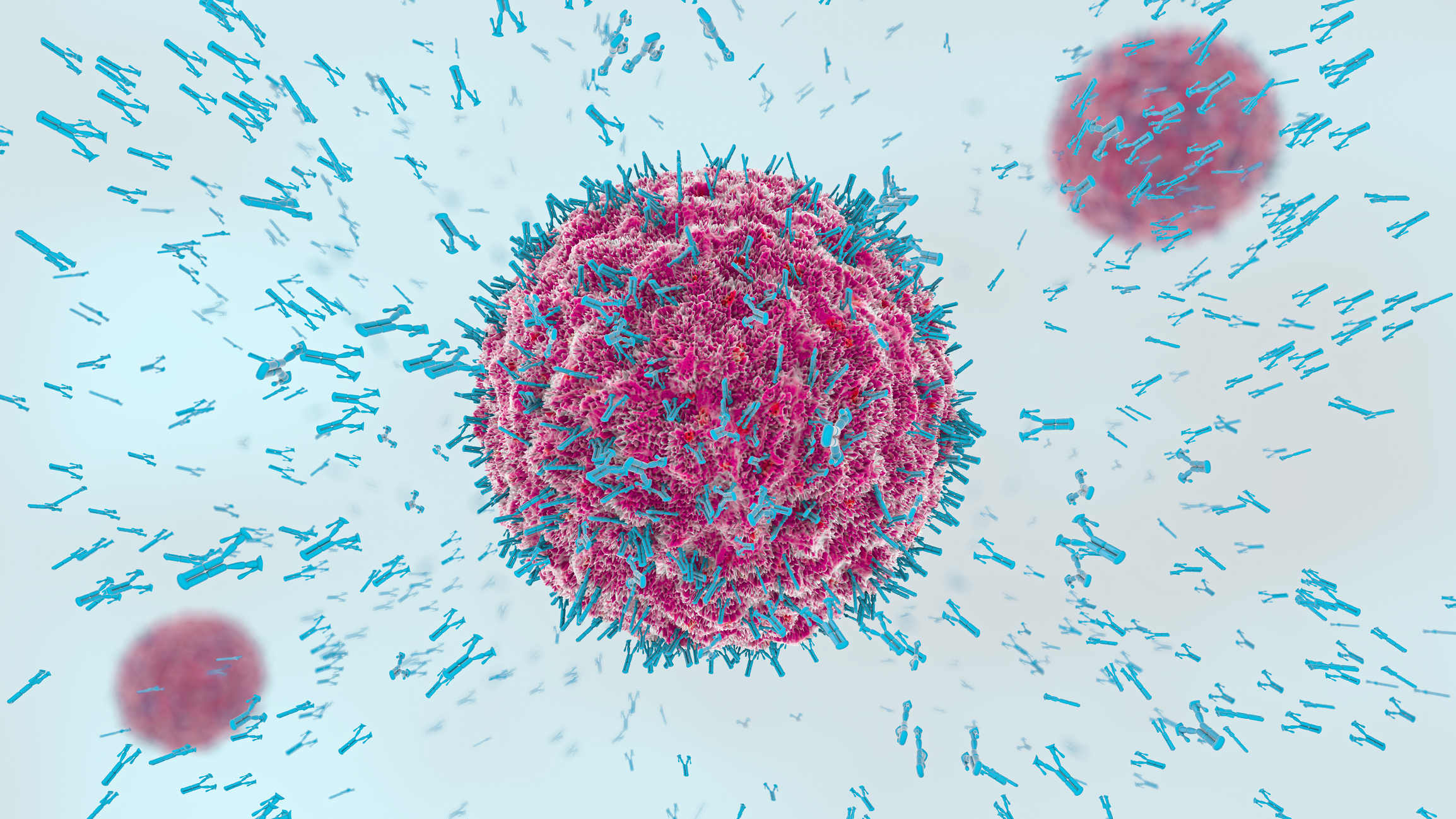
The ability to isolate and identify proteins from a biological solution is fundamental to basic research and clinical diagnosis. Proteins are the workhorses of the organism; they send and receive messages, they control the flow of information across the cell membrane, and they enact cascades of action within cells. It is rare that a single protein works alone, so it is imperative to understand how proteins interact with each other if we are to understand the nature of our bodies and to discover and treat disease.
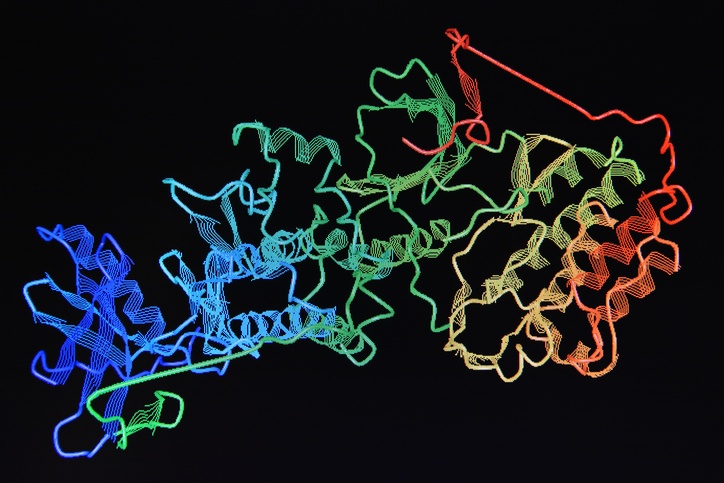
Modern drug discovery utilizes libraries of purified proteins. These proteins are screened by libraries of small molecule drug precursors. This combinatorial screening process greatly speeds up the identification of new drug molecules.
Size exclusion chromatography columns are used to separate molecules by size, molecular weight, and hydrodynamic volume. The technique can be used with proteins, polymers, and other macromolecules. It can also be used for buffer exchange or desalting a sample. The principle behind size exclusion chromatography columns is simple, but the technique only works when the correct resin-bound column is matched to the experimental goal.

Nano gold is another name for gold nanoparticles. These nanoparticles are a fraction of the size of human hair and are less than 100 nm in diameter. Nano gold particles are so small that it they are generally found as a colloidal solution, which means that the gold nanoparticles are suspended in a liquid buffer. Therefore, nano gold, or gold nanoparticles are also called colloidal gold. Also, nano gold is generally found in a colloidal solution because gold nanoparticles are created by citrate synthesis. This process involves mixing solutions together to result in the precipitation of gold nanoparticles into solution.

From the clinical laboratory ELISA to the home pregnancy test, the conjugated antibody is integral to the function of many diagnostic assays. We know about the specificity of antigen-antibody interactions and their role in mounting the innate immune response to a pathogen. This antigen recognition specificity has been utilized by clever scientists and engineers to create biosensors capable of detecting the presence of antigens in biological samples. Biosensors come in many shapes and sizes, and have varying levels of complexity, but one fundamental concept is the need to covalently attach antibodies to a substrate.

The China Association for Chemical Laboratory Practice (CACLP) Expo is the biggest and most influential IVD-exhibition in the Asian giant. We attended the 2018 edition, held in the city of Chongqing from March 17th to March 19th. Here are the impressions about the event of some colleagues and our CSO.
The last two decades have seen an explosive growth in the use of magnetic beads in Life Science, with sustained double figure sales increase all across the industry. The main driver of this success has been the use of magnetic beads as a solid phase on Chemiluminescence Immunoassays (CLIA) kits. Thanks to its easy automation, this technique has become the preferred choice for high throughput In Vitro Diagnostic.
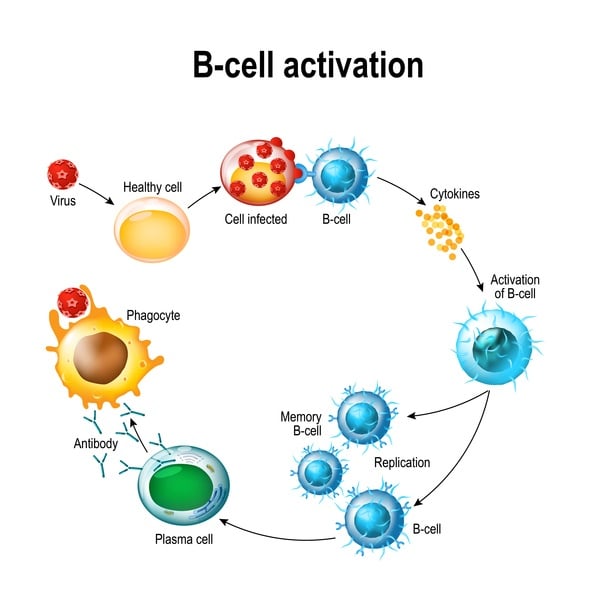
The binding specificity between antibody and antigen drive our immune systems to successfully fight infection. When a viruses or bacteria invade a body they are engulfed by macrophages, which break them down and present their epitopes to the B cells lymphocytes. These B cells read the epitope and create antibodies with an antigen binding site, or paratope, that specifically recognizes the invading pathogen, binds to it, and signals to the rest of the immune system that the pathogen/antigen should be destroyed. This antibody affinity to antigen is similar to the specificity of a key in a lock.
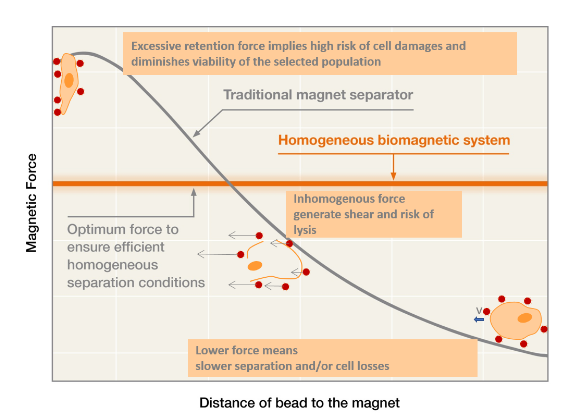
Magnetic beads and separators have grown in popularity as a biotechnological tool over the past few decades. As we gained experience with the technology, we learned that all magnetic separators are not created equal. It turns out that it isn’t just the magnitude of the magnetic force that improves separation time, it is the way it spreads out over the working area that matters the most. A homogeneous magnetic separator is designed to ensure that every point within the working volume experiences the same magnetic force. This is especially important when scaling up a biomagnetic separation process from milliliters to liters.

The collection of tissue is a fundamental procedure for research and clinical biology. Before collection it is important to consider which method will be used to preserve the tissue and prepare it for histology or molecular analysis. There are two options to choose from when collecting and preserving tissue: frozen or formalin-fixed paraffin-embedded (ffpe). Each has its strengths and weaknesses, but these are only apparent when the intended use of the tissue is considered.
Anti human IgG is the most prevalent antibody isotype in human serum. It plays a critical role in the adaptive immune response. When a pathogen invades a human body it is intercepted by a white blood cell called a B-cell. These B-cell lymphocytes have cell surface receptors that bind a wide array of pathogens. Once activated, the B cell divides and produces offspring cells which secrete very specific antibodies capable of identifying that exact invading pathogen. These antibodies recognize a specific antigen binding site.

The synthesis of complementary DNA (cDNA) is fundamental to the study of RNA expression in cells and tissues. RNA is fragile and not compatible with the polymerase chain reaction (PCR). Therefore, an intermediary step is needed to convert RNA to cDNA for analysis by quantitative PCR (qPCR). This qPCR technique utilizes specific primers and polymerase enzyme to amplify targeted sequences of cDNA. Since the cDNA is complementary to the initial mRNA isolate, this technique provides a quantitative readout of mRNA levels in the sample. The need for cDNA is so great that many companies sell cDNA synthesis kits to simplify the procedure.
We are all familiar with magnetic materials, but not so much with what exactly ‘magnetic’ means. The magnetic moment, usually quoted as ‘m’, it is defined as the property that makes a material align with an external magnetic field.

A CHIP assay is short for Chromatin Immunoprecipitation assay. This technique is used to gain insight into the region of the genome a particular protein is associated with. To do this the CHIP assay captures information about the protein-DNA interactions in the histones of genomic DNA. The protein-DNA binding structure is preserved because the histone is crosslinked before the cell is lysed. The protein and DNA are bound together even after the chromatin is broken up into smaller pieces. Then the protein-DNA conjugates are captured by immunoprecipitationwith antibodies binding specifically to regions in the protein of interest. Then the DNA is extracted from the proteins and analyzed by targeted qPCR or genome-wide next-generation sequencing (NGS).

Biotinylation is the process of attaching a biotin tag to a molecule. The molecule can be a protein or an oligonucleotide. Biotinylated molecules are useful in many biological contexts, but are primarily used for capture or detection of target molecules. Biotinylated molecules are used in Western blotting, ELISA, flow cytometry, and other inventive detection methods.

Filtration is a common technique for mechanically separatinggases or liquids. A filter membrane serves as a barrier to enable ion exchange, removing macromolecules or bacteria from solution, separating colloids, or recovering gases. Some uses for filtration include producing clean drinking water, generating safe food products, and ensuring a clean environment. It is used for petro-chemical vapor recovery, oxygenation of blood in an artificial lung, and hemodialysis via an artificial kidney. Filter membranes are necessary for ion exchange in a fuel cell and for electrolysis. Filtration is also a key procedure in the laboratory to separate RNA, DNA, cells, proteins, and other molecules.
Common methods include microfiltration (0.1 to 1 micrometer particulates), ultrafiltration (0.001 to 0.1 micron), nanofiltration (less than 0.001 micron), and reverse osmosis (less than 0.001 micron). Microfiltration is used to separate colloidal solutions, but the other three are used for dissolved particulates.
Enzyme immobilization onto solid supports is a convenient way to control and reuse enzymes. The enzyme is a catalyst for a reaction, meaning that it enables or speeds up the chemical conversion from reactants to products. This is a valuable property for both small scale research and large scale industry including biotechnology, pharamaceutical applications, food production, and wastewater treatment. There are many materials used for immobilized enzyme solid support systems, and a variety of enzymes can be used. Perhaps the most advantages solid support system is one that is magnetic. A magnetic bead is easily recoverable with the application of an external magnetic field. This ensures that reusing the enzyme is quick and easy.
The traditional biomagnetic separation is a permanent magnet block. The test tube is placed next to the magnet and the magnetic particles in solution move toward the magnet. This system works for very small volumes, but it is not the most efficient method and problems often arise in larger volumes. The downfall of this geometry is that the permanent magnet is only on one side of the tube, which means that the magnetic particles are only drawn to that one side. The magnetic particles close to the magnet will experience a higher force than the magnetic particles farthest away from the magnet. Magnetic force decreases as distance from the magnet increases, so the particles farthest away might not feel any force if the magnetic strength is not great enough.
Centrifugation is a common technique used for the separation of heterogeneous mixtures. The force of gravity on matter is the core principle of centrifugation. Matter naturally separates based on density, with the most dense particles precipitating out of solution first, and the less dense particles falling out later. Some particles prefer to remain in colloidal solution under normal gravitational force, and will not separate naturally within a reasonable time frame.Centrifugation can be used to rapidly separate those mixtures. The spinning centrifuge creates centripetal forces much greater than gravity, which can force particles to separate. The centrifugal protocol must be optimized for each experiment in order to efficiently separate the mixtures into desired layers. The most basic separation results in a higher density pellet at the bottom of the tube, and a lower density supernatant. Differential centrifugation is a process whereby the pellet and supernatant are separated during multiple centrifugation steps. Density gradient centrifugation utilizes a density gradient matrix within the tube to aid in separation. Another useful tool for centrifugation is a centrifugal filter. The filter helps to separate particles by size as well as density in one swift motion. The centrifugal filter is used to isolate RNA or DNA, to consolidate proteins, to separate molecules by size, or to remove contaminants from a liquid.
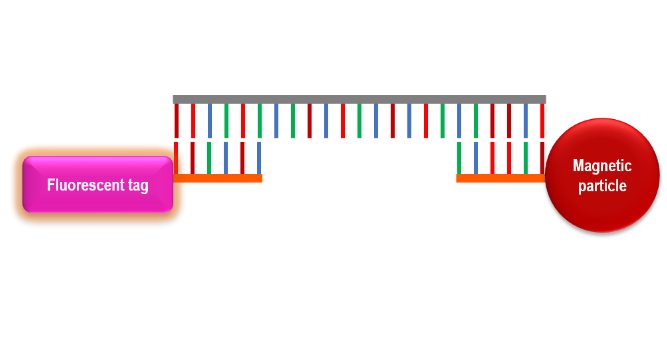
The reversible-binding systems mentioned in the previous post are examples of non-specific capture methods. They capture total DNA and RNA in a sample because they simply rely on the affinity of nucleic acids to the magnetic particle coating or functional moiety. Non-specific capture methods bind all single-strand (ssDNA) or RNA regardless of sequence. A more specific capture system is able to target specific sequences of ssDNA or RNA. This type of specific capture is most applicable to diagnostic systems as an assay for a specific pathogen. Examples include systems to diagnose methicillin resistant staphyloccocus aureaus (MRSA), specific plant viruses, and infection with malaria-causing bacteria. These strategies could be applied to detect viral, bacterial, or fungal pathogens in a variety of clinical samples.

Cleaning validation is an important regulatory component of industrial manufacturing. It ensures that a standard and reliable protocol is followed forthe removal of residual chemicals from equipment between batches. Clean equipment is important for process validation because consistent products cannot be synthesized without consistent starting points. However, cleaning validation is about more than just equipment cleaning. Attention must be paid to other parts of the manufacturing process including employee education, building and facility maintenance, equipment calibration and maintenance, control of raw materials, up-to-date standard operating procedures, clear communication between all employees, and accurate reporting of data and processes.
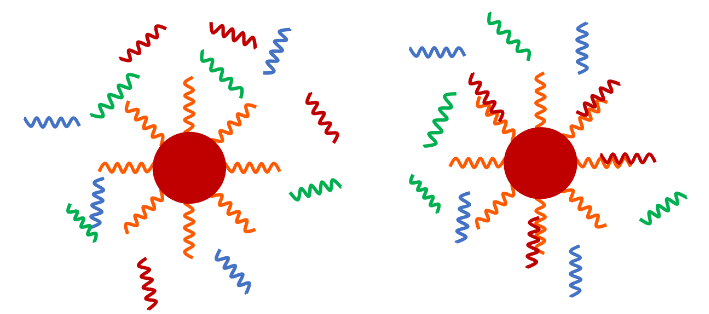
The coating and surface chemistry of magnetic beads governs the binding efficiency of target DNA or RNA. Superparamagnetic beads for life science applications come in two general forms: core-shell type and embedded type. The core-shell synthesis method produces beads composed of a single superparamagnetic core with a polymer or silica surface coating. One example is a bead composed of a magnetite core surrounded with a dextran shell. Other beads of the core-shell type are composed of a polystyrene or polyvinyl alcohol (PVA) core surrounded by superparamagnetic particles and protected by a surface coating.
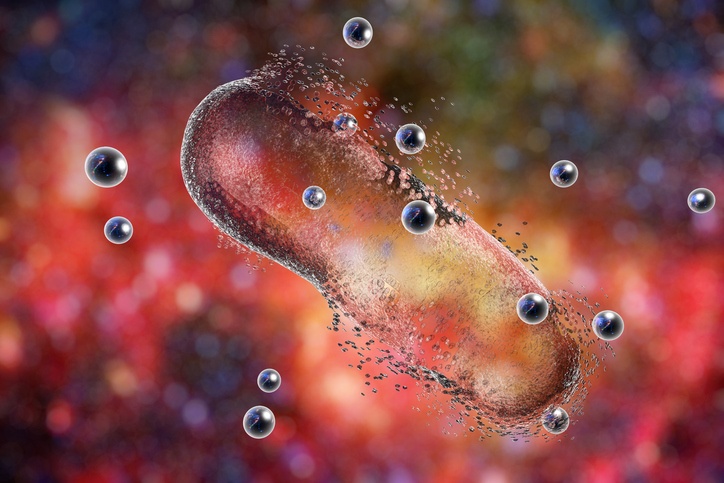
Superbugs, or bacteria that are resistant to currently available antibiotic treatments, are of growing concern to human health worldwide. Many of these superbugs are present in hospitals, and are frequently colonizing surgical sites and causing life-threatening infections and sepsis. The presence of these bacteria in blood is commonly detected by traditional culture methods that require one to two days. There is a need for a faster method to identify the presence of the bacteria and to take measures to prevent the spread of infection as rapidly as possible. One proposed technology is to use immunomagnetic separation with conjugated fluorescent probes to selectively bind bacteria and quickly visualize their presence in whole blood samples. Recently, fluorescent magnetic multifunctional carbon dots have been coupled with superbug-specific antibodies for the identification of the drug-resistant superbugs Staphylococcus aureus (MRSA) and Salmonella enterica serotype typhimurium definitive phage type 104 (DT 104) present in whole blood samples.
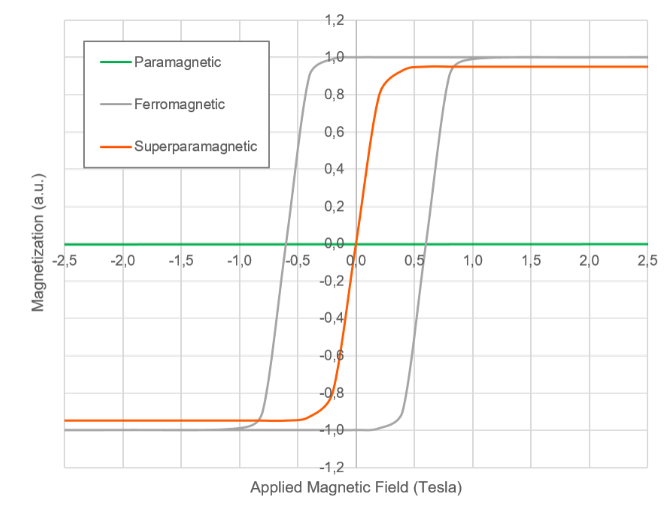
Magnetic DNA or RNA purification relies on the superparamagnetic property of micro- or nano-scale particles. These particles are most often made of iron oxide, with magnetite (Fe3O4) more commonly used than maghemite (Fe2O3). Superparamagnetic particles are not innately magnetic, but they become magnetized when influenced by a magnetic field. So, if the magnetic field is zero, then the particles are not magnetic at all, but when a magnetic field is applied the particles become magnetized.
The traditional solid-phase support system is a static column. These columns are made of silica matrices or anion-exchange resins. They are porous and allow the solution to flow through them. These non-magnetic solid-phase columns require centrifugation to force the solution through. More recently, magnetic particles are being used as mobile solid-phase support systems for capture and purification of DNA and RNA. These magnetic particles are added to solution and are free to move around during the DNA or RNA adsorption period. They are then retrieved by magnetic separation. No centrifugation is needed for magnetic-particle nucleic acid purification.
Nucleic acid separation can be fickle. DNA is fragile, and RNA even more so. Many commercial kits are designed to streamline the process, but they may not result in high yield or high purity DNA or RNA every time. Every laboratory is different; working habits vary, and experimental goals are not identical. It is tempting to rely on a single kit for routine isolation of genetic material because it is familiar, your lab may have published previous work with an established protocol, or you might not have the luxury or freedom to try something new. However, if you are experiencing consistently low RNA or DNA yield or purity then you may be able to justify taking time to gain a deeper understanding of the process, familiarizing yourself with the tools available, and possibly reworking your strategy.

Biomagnetic separation is a versatile and widely used tool in both industrial and small laboratory settings. It is used for the isolation of target drug molecules in the pharmaceutical realm, for the enrichment of enzymes in industry, and for in-vitro diagnostics in medicine. It is especially useful in the small-scale research environment for inexpensive target cell enrichment, protein isolation, or nucleic acid capture. In the early days of biomagnetic separation it was thought that the process was only reliable for small volumes. However, the development of modern biomagnetic separation racks has made it possible to scale up the process to large volumes and to enable process validation and consistency between batches.

The ability to obtain an enriched population of small molecules, cells, proteins, nucleic acids, or contaminant-free solutions is important for all applications: from small laboratory research up to the large-scale production of pharmaceutical products. Filtration systems are available in all shapes, sizes, and materials for diverse situations. The need to meet regulatory guidelines for purity and consistency of pharmaceutical productsdemands a well-designed enrichment plan and filtration system.

Janus particles named after Roman god Janus who was illustrated with two faces, one looking to the future, and the other to the past. Pierre-Gilles de Gennes mentioned the term Janus particle in his Nobel lecture on soft matter in 1991. Janus particles are synthesized by joining two or more different materials together. Each material imparts a unique and useful quality to the particle; therefore, one Janus particle can display multiple properties and functionalities. It is this fundamental asymmetry that makes Janus particles a powerful tool in biotechnology for biosensing, bioseparation, self-assembly, drug delivery, and much more. This blog post will summarize a lengthy and informative reviewarticle of the same title that was published in Chemical Reviews in 2013.

Janus Particles and anisotropy
Janus particles (JPs) have two or more properties or functions wrapped up into one package. There are many different synthesis methods, properties, and applications for JPs. Anisotropy is defined as an object having a different physical property or different value when measured in different directions. Janus particles are anisotropic. Their properties are asymmetric. They can have a hydrophobic face and a hydrophilic face. They can have magnetic properties as well as optical reporters on the same particle. JPs can have different optical properties on either side. They can have different ligands on opposing faces. It is anisotropy that makes them useful tools for biosensing and bioseparation. One particle with multiple functionalities is often more useful than an isotropic particle with one property.

Antibodies labeled with fluorescent tags, radioactive molecules, biotin, enzymes, and other small molecules are essential for immunoassays, cell sorting, diagnostic assays, cell imaging, and other procedures. The attachment of small molecules to individual antibodies is not trivial, and to do it with a specific orientation is especially challenging. The purified antibody to be labeled must be in solution at high concentrations, and the process requires multiple washing steps. On-bead labeling methods are used to improve the labeling efficiency. Protein A binds to the Fc region of IgG antibodies across a variety of species, and is commonly used to capture antibodies onto a bead surface for isolation or for small molecule modification.

Streptavidin magnetic beads are used to isolate biotinylated biomolecules
The affinity between streptavidin and biotin is one of the strongest non-covalent interactions known in nature. It is extremely specific and is unperturbed by extreme changes in temperature, pH, and detergent. It is therefore a very useful tool in biotechnology. For example, the system is commonly used in magnetic separation. Target molecules are biotinylated, which means that a biotin molecule is covalently attached to the protein, nucleic acid, or other molecule of interest. Streptavidin-bound magnetic particles are incubated with a solution containing the biotinylated target molecule for a sufficient time for the streptavidin-biotin affinity to form. The complexes can then be isolated from solution by magnetic separation.

The Zika virus (ZIKV) is emerging as a global public heath threat. It has been linked to the development of microcephaly in newborn babies whose mothers’ were infected by ZIKV during pregnancy. Recently, it has been shown that the virus can be sexually transmitted. The clinical signs in adults include fever, rash, joint pain, and conjunctivitis, which are common clinical signs for a general viral infection. This makes clinical diagnosis difficult without a specific immunoassay. The current methods for ZIKV detection are qPCR (quantitative polymerase chain reactions) or ELISA (enzyme-linked immunosorbent assay). Each of these is time consuming and the reagents can be expensive. As more people become infected and the virus spreads throughout the population it will become necessary to find a cheaper and faster alternative assay for detection of ZIKV. A magneto-actuated chemiluminescence immunoassay for the Zika virus has been developed. The demonstrated limit of detection is comparable to qPCR.
CD4+ lymphocytes are of particular interest in patients infected with Human Immunodeficiency Virus (HIV). A devastating effect of this retroviral infection is a progressive loss of CD4+ cells. These cells are crucial to a healthy immune system. When the level of CD4+ cells fall below 200cells/uL whole blood the patient is diagnosed with Aquired Immune Deficiency Syndrome (AIDS).
Therefore, the number of CD4+ cells in these patients is a good way to determine when to start antiretroviral treatment, and to administer drugs to protect against opportunistic pathogens. Currently, the most accepted way to measure the CD4+ concentration in whole blood is to use flow cytometry. However, this technology is extremely expensive and requires trained technicians to operate and maintain the machine. An alternative method is needed in resource poor areas. This alternative method would ideally be implemented at the point of care to avoid transportation of samples to and from a laboratory. One such recently developed technology is a magneto-ELISA to detect the concentration of CD4+ lymphocytes in whole blood. The detection system relies on magnetic separation to obtain an isolated population of CD4+ cells. It has a demonstrated limit of detection of 50 Cd4+ cells per μL, which is sensitive enough to diagnosis AIDS.

Efficient methods for DNA detectionin clinical, environmental, and experimental samples are constantly in demand. In a clinical sample, DNA capture and identification can be essential to the diagnosis of disease. In public heath and environmental situations it can be used to identify contamination of food or water. DNA collection and characterization is constantly expanding our ability to answer experimental questions. DNA capture is a mainstay of modern biotechnology. Traditional techniques rely on affinity columns, centrifugation, and multiple washing steps. Newer methods are based on magnetic separation. In the beginning, magnetic separation was limitedto packing a column with magnetic material and running a solution through it. As nanotechnology evolved it became possible to use mobile solid support systems such as magnetic nanoparticles.
When food becomes contaminated with pathogens it is necessary to identify the contamination and remove that food from the supply line as soon as possible. When that food is perishable it becomes all the more important to act quickly, but this demands a rapid detection system. This “need for speed” automatically eliminates the traditional culture method from the list of potential detection techniques. A commonly used method for pathogen detection is to isolate the pathogen and concentrate it enough to be accurately amplified and detected by real time quantitative polymerase chain reaction (RTqPCR). However, the method used to capture and concentrate the pathogen needs to be rapid and efficient. Magnetic nanoparticles and magnetic bead separation techniques are the perfect technologies for the rapid isolation and concentration of pathogens.
The bacteria Staphylococcus aureusis of major concern in modern healthcare.Once it spreads to a patient’s bloodstreamthe infection can lead to sepsis and death. Hospitalized patients are particularly vulnerable, especially those with weakened immune systems. Some strains of S. aureushavebegun to demonstrate antibiotic resistance. For these reasons it is important to identify and contain infection early; rapid detection of bacteria in patient sera is key. Traditional culture methods are time-consuming and require transportation of samples to offsite clinical laboratories.Development of a rapid point-of-care diagnostic tool is desirable, andthe magnetic nanoparticleis rapidly becoming the tool of choice.
First use biomagnetic separation to isolate the target molecule
Magnetic nanoparticles are fantastically customizable due to surface coatings and functional moieties, and are easily recoverable from solution. A functionalized nanoparticle will bind to its target after a short incubation time, and the conjugate can be isolated with the help of a magnetic separation rack. This technology allows for the rapid selection and concentration of a target molecule. Once the target molecules, in this case S. aureus bacteria, are isolated, further identification methods can be used. This is where a group of researchers working in Istanbul, Turkey became creative.
The grouphas engineered a S. aureus detection system using a mesoporous silica nanoparticle-oligonucleotide conjugate. The system’s limit of detection is 682 cells/mL, which indicates potential for use as a point-of-care diagnostic. The system detects Micrococcal nuclease (MNase), which is an enzyme specifically secreted by S.aureus. MNase degrades RNA or DNA, and is well known within the field to be a marker of S. aureus infection.
The system works as follows:
- A sample of blood (1 mL) is mixed with magnetic nanoparticles coated with silica and functionalized to bind to S. aureus.
- The nanoparticle-bacteria conjugate is isolated from solution by biomagnetic separation.
- The isolate is heated to release MNase out of the bacteria and into solution
- A non-magnetic mesoporous silica nanoparticle filled with fluorophore is added to the solution. The pores of this nanoparticle are sealed shut with oligonucleotides that are resistant to degradation by all nucleases except MNase. When the pores are capped the fluorophore is quenched and no fluorescence is visible.
- If MNase is present in solution the oligonucleotide caps are degraded, the nanoparticle pores open, and the flurofores are released. The free fluorphores are no longer quenched, and a fluorescence signal is measured.

The use of magnetic nanoparticles and biomagnetic separation to isolate proteins from solution is advancingover more traditional column-based methods. Magnetic nanoparticles provide a gentler and more rapid separation process compared to column-based methods, and often demonstrategreater yield. With a properly scaled separation rack the separation process can be monitored in real-time and calibrated across experiments.

There are many variables to consider when designing a magnetic separation system and tailoring it to the experimental goal. Bead surface functionalization is one key component, and often receives the most attention because it is the most obvious point of control for assay specificity. However, it is easy to lose site of the fundamental mechanics of magnetic separation. When designing a magnetic separation protocol it is also important to consider the movement of the magnetic particles in the magnetic field gradient. Superparamagnetic particles will form chains when placed into a magnetic field, a key property to have high enough separation speed for practical purposes.
Biomagnetic separation is a technique that uses small superparamagnetic iron-oxide particles and a magnet to separate cells or nucleotides from solution. These particles are often called magnetic nanoparticles or microspheres due to their small sizes ranging from less than 100 nm to 5 μm. The magnetic particles are chemically altered (functionalized) to be biologically compatible and to specifically bind to a variety of different cell types. The targets of interest can be bound by the particles and isolated from solution (positive selection) or the unwanted targets can be bound and removed from solution (negative selection). Magnetic bead cell sorting has many advantages over traditional column-based methods due to increased specificity, decreased procedure time, and increased cell viability.
Immunomagentic separation is a selective and faster assay than microbiological culturefor detection of bacteria in food, water, or clinical samples, but it still has limitations. A novel bacteriophage-based magnetic separation has been developed to take the magnetic separation assay to the next level.

The study of protein complexes is often difficult due to their physical properties. Proteins are many times hard to isolate with traditional chromatographic methods, especially large molecules in complexes.This is due to most complexes being maintained by non-covalent bonds, which are easily disrupted during isolation.
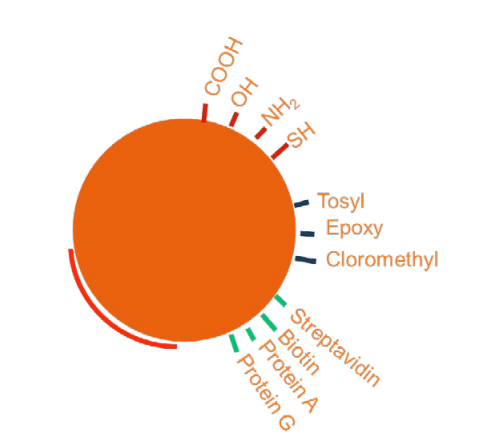
Magnetic bead technology has developed rapidly in the past decade. New functionalization strategies are continually hitting the market as more laboratories begin to use magnetic separation systems to identify and isolate cells and microorganisms. There is a direct correlation between advanced functionalization strategies and improved assay selectivity. The size of magnetic beads (μm-nm) places them squarely into the cellular realm, and their surface functionalization causes them to bind to specific surface ligands. This functionalization step is crucial to the separation process because it allows the operator to control which cells the beads will bind to. For a positive-selection immunoassay, the bead surfaces are functionalized by covalent attachment of an antibody that will bind to the cell or micro-organism of interest. This bond ensures that the microbe-bead conjugate will be captured in a magnetic field gradient.

Within Nanoparticles (NP), Magnetic nanoparticles (MNPs) can be influenced using an applied magnetic field. Due to restricted size effects such as high surface-to-volume ratio, these MNPs demonstrate high magnetic properties compared to their corresponding large particles. The component of magnetic nanoparticles ranges from 5.0 nm to 50 nm in size and display a unique feature known as ‘super magnetism’ in the presence of an externally applied magnetic field.
Magnetic beads or Magnetic labels are the most suitable nanometric-sized iron oxide particles for extracting biological molecules prior to their detection. These are either encapsulated or attached together with polymers with the size ranging from 35nm to 4.5μm. First discovered by French scientist Louis Neel, a winner of Nobel Physics Prize in 1970; these magnetic labels exhibit several features, well tailored for various applications.
Vacuolar processing enzyme (VPE) is involved in manyplant physiological processes and functions as a cysteine protease. It plays important roles in immunity, programmed cell death, leaf senescence andseed maturation. VPE has caspase-1-like activity and it is thought that caspases are evolutionarily related to this enzyme. The biological relevance of VPE makes the development of tolls to study this enzyme extremely valuable for research purposes.
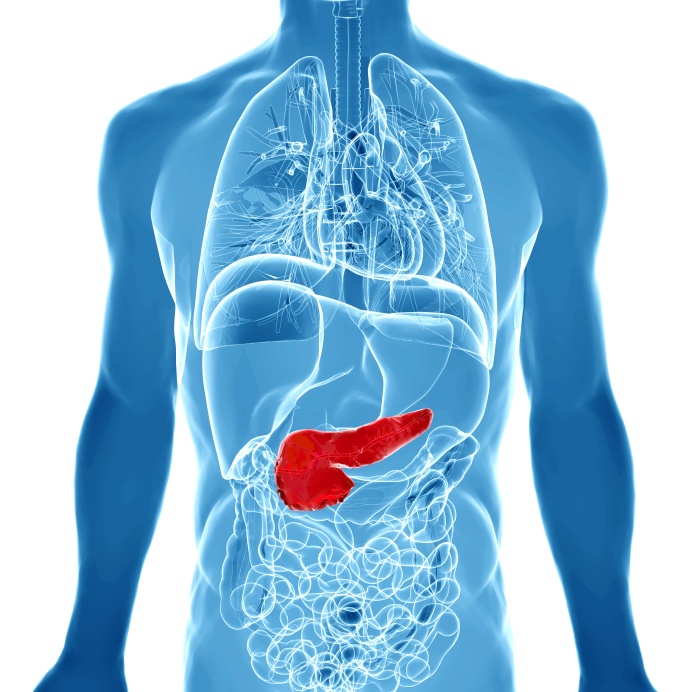
Pancreatic cancer is one of the most deadly forms of cancer with a survival rate of only 6%. Most cancer therapies are not effective in the treatment of pancreatic cancer because cancer cells are intrinsically resistant. The study of molecular and cellularcharacteristics of the tumor is therefore essential to understand how this cancer works at the molecular level and to develop new therapies. We now have all the techniques available to do a full characterization of cancer cells, such as DNA and mRNA sequencing, and signaling protein arrays. This molecular characterization allows a better classification of the tumor and therefore helps to predict response to treatment and prognosis.

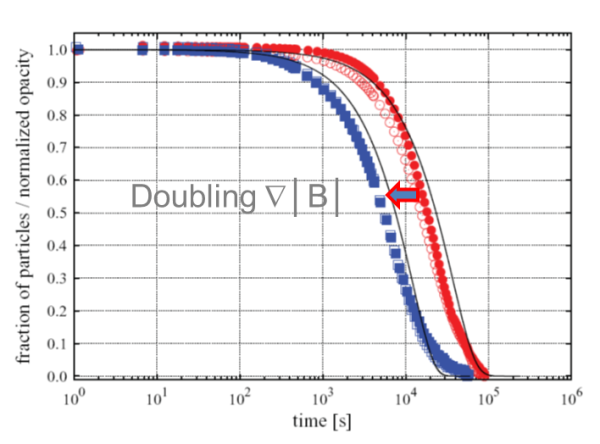
The scaling up of a biomagnetic separation process is not an easy task if we use classical magnetic separators. In these devices, the magnetic force over the beads changes with the distance, thus the magnetic force profile is very different at different volumes. However, using advanced biomagnetic separation systems, the magnetic force becomes constant at all the working volume. Then the process scaling up is straightforward: you just need to use systems with the same magnetic force, and you would have the same separation conditions regardless the scale.
Mycobacterium avium subsp. paratuberculosis (MAP) is the causative agent for Johne’s disease that affects cattle and leads to wasting of the infected animals. In a herd, most animals infected are asymptomatic but still propagate diseasethrough their milk and feces. This makes the most susceptible animals develop sickness and the within-herd transmission uncontrolled. It is therefore important to detect MAP infection early and even when it is causing only latent infection. The most used test for the detection of MAP in animal blood or milk samples is ELISA with MAP-specific antibodies. The problem with this essay is that the detection limit is high and this leads to false negatives, which limits the effective detection of infected animals. The most sensitive method available is liquid and solid culture. However, since MAP has a slow growth rate and no media is truly selective for these bacteria, there are a lot of instances where culture overgrowth by contaminating organisms impairs MAP viability and limits detection to 23% with many false negative results. Molecular based methods such as PCR are also used and are more rapid and sensitive but they do not assess the viability of MAP cells.
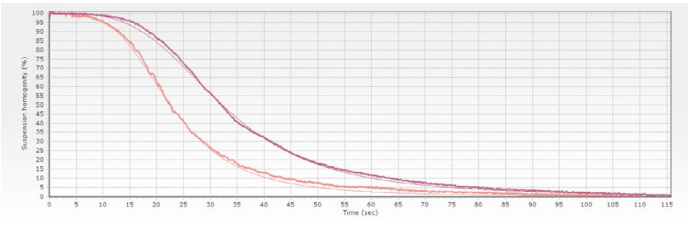
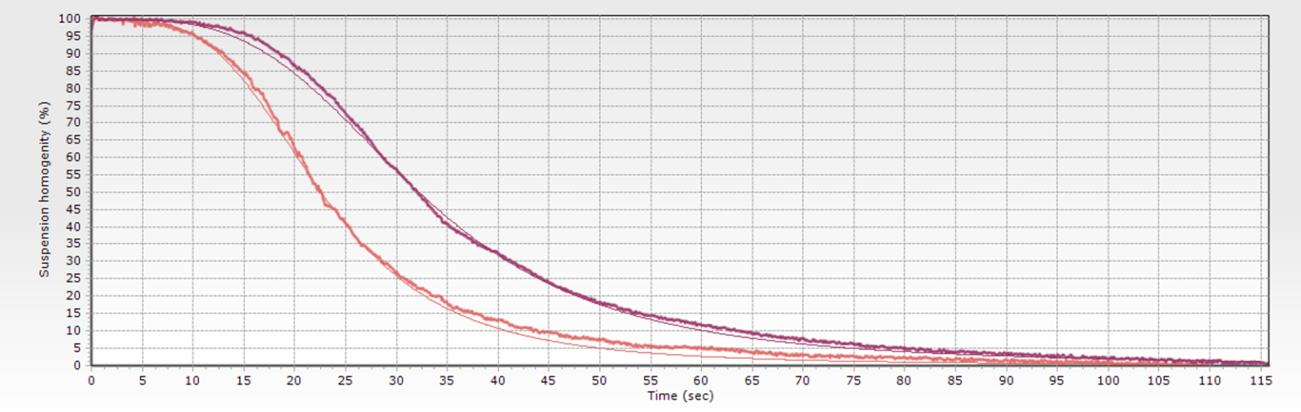
Stablishing a separation time is the most usual way to specify a biomagnetic separation process. The problem is that this magnitude is not only related with the magnetic beads suspension, but also with the specific system used to perform the separation.

Nucleic acid detection is crucial in the clinical practice since it can be used to rapidly identify pathogenic organisms. Rapid detection with highly sensitive methods is essential for diagnosis and choosing treatment courses. This is a complex task since samples need to be enriched with the nucleotide targets and at the same time needs to eliminate nucleases, PCR inhibitors and host material. This means that to detect and identify genetic material from a pathogen, in the form of 18S and 28S rRNA, in an efficient and reliable way, the protocol needs to eliminateeukaryotic rRNA from the sample to avoid competition for the substrates during the cDNA synthesis, since it decreases the detection sensitivity.

The Sepmag equipment is mentioned in the Encyclopedia of Inorganic and Bioinorganic Chemistry on the chapter “Biomedical applications of magnetic nanoparticles” written by Daniel Ortega. This chapter starts with examples of several magnetic isolation methods such as the isolation of macromolecules from enzyme separation that involves immobilized affinity ligands to the removal of cancer cells frombone marrow, which is one of the most important applications ofbioseparation techniques.

Magnetic separation techniques have become an essential tool in many areas of research. Most technological advances in these techniques have been possible due to the work of many researchers trying to solve everyday technical problems or optimizing existing protocols. In order to get the specialists in the field from all over the world together, meetings are an important way for contact between researchers that can help find new applications and solutions in the scientific and clinical setting.

Renewed version of ‘The Basic Guide to Scale-Up Biomagnetic Separation Processes’ e-book is already available (FREE)!

Magnetic separation techniques are becoming more and more versatile as researchers take advantage of this technology to tackle everyday problems and come up with innovative solutions. One striking example of this innovation capacity is the technique developed recently by Ziyuan Wang and colleagues. These researchers combined magnetic separation beads with bacteriophages to selectively isolate bacteria. Since they wanted to purify Escherichia coli from samples with high affinity and specificity they used T7 bacteriophages to act as binding molecules, similarly to how normally antibodies are used.
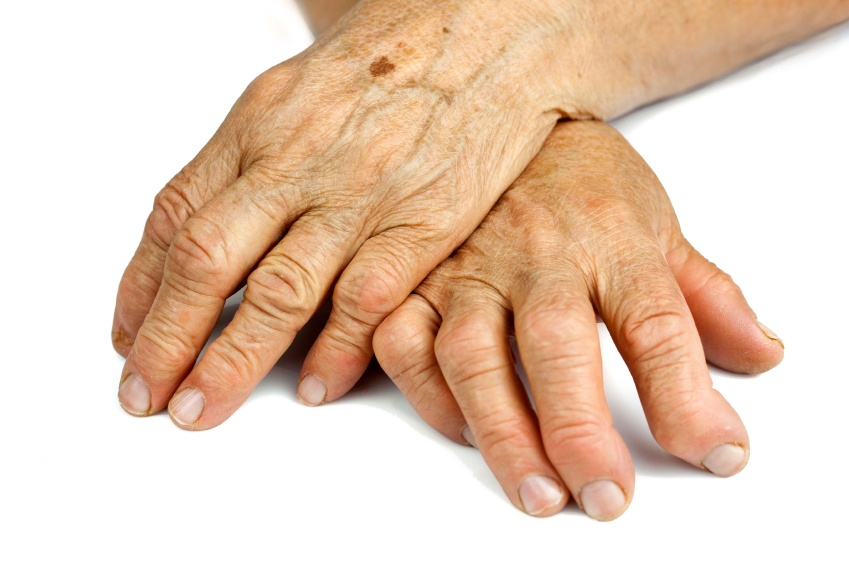
Clinical validation of Raman scattering with magnetic beads in the diagnosis of rheumatoid arthritis
Surface-enhanced Raman scatteringis a new technique to perform immunoassays that can be used to detect several disease biomarkers.It is better than traditional immunoassays, like ELISA, since it is more sensitive, selective and takes less time, which are precious things in diagnosis.

Uranium is an environmental pollutant with severe health and ecological consequences since it has radioactive and chemical toxicity affecting mammalian reproduction and development. Therefore, the environmental detection of this element is essential, to prevent deleterious effects on the ecosystems.
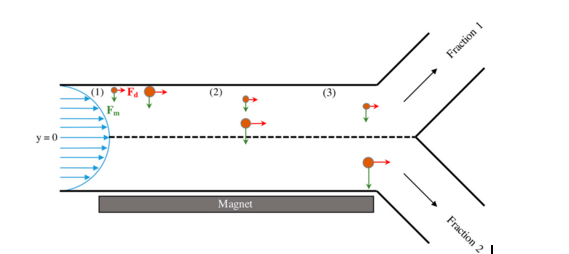
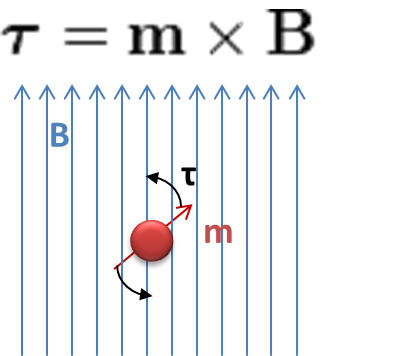
Magnetic nanoparticles are susceptible to a magnetic field. If the magnetic field is constant then the particles will experience torque and will spin in place
Τ = m x B

The size of magnetic nanoparticles is very important. The behavior of the magnetic particle in a magnetic field gradient is dependent upon the particle’s diameter. The ability of the particle to overcome drag force and move in the direction of a magnetic field gradient is dependent on the size of the particle.
Extracellular vesicles are structures released by cells and include exosomes,microvesicles, and apoptotic bodies. These vesicles are an important diagnostic toll since they carry substances from their cells of origin into circulation. The biomolecules contained within, such as RNA and proteins, can be used to determine if the cells that produced them are being affected by a disease. For example, cardiovascular diseases and cancer, lead to increase in specific extracellular vesicles. Isolating these vesicles form body fluids is not a simple task.
The study of biological processes is getting more complex and has to take more factors into account. Physical forces are an important part of normal physiological processes and their impact has been largely overlooked in the in vitro study of biological mechanisms. The cellular force sensing mechanisms play important roles in disease. For example, in atherosclerosis endothelial cells have to respond to pulsatile forces and stiffening of the arteries.
Repair of blood vessels is essential to maintain homeostasis and this process is driven endothelial colony-forming cells. These are vascular lineage-specific progenitor cells that have the ability of driving post-natal vasculogenesis. Endothelial progenitor cells are mobilized into circulation by mediators released due to vascular trauma. Initially, it was though that these cells only acted during embryonic development, but now we know that they are equally important to restore the tissues’ vascularization after disease, especially in heart disease and cancer. Therefore, these cells have therapeutical potential in the treatment of conditions affecting the vascular network. The problem is that these are very rare cells in circulation accounting for only 0.01% of all cells.
Connective tissue progenitors (CTPs) are used as a therapy for bone repair. These cells can be isolated thanks to their expression of hyaluronan, a membrane glycosaminoglycan. Since these cells are rare relatively to other cells in the bone marrow (1 for 20000) it is difficult to obtain enough cells for therapeutical application. The problem with the application in humans is that CTPs need to be isolated from bone marrow suspensions to obtain a high number of viable cells. This procedure implies collecting bone marrow, separating the CTPs and engrafting them in the patient which, due to the complicated nature of rare cell isolations, would need two surgical procedures. Therefore the process of separation needs to be quick and efficient without impairing cell function.
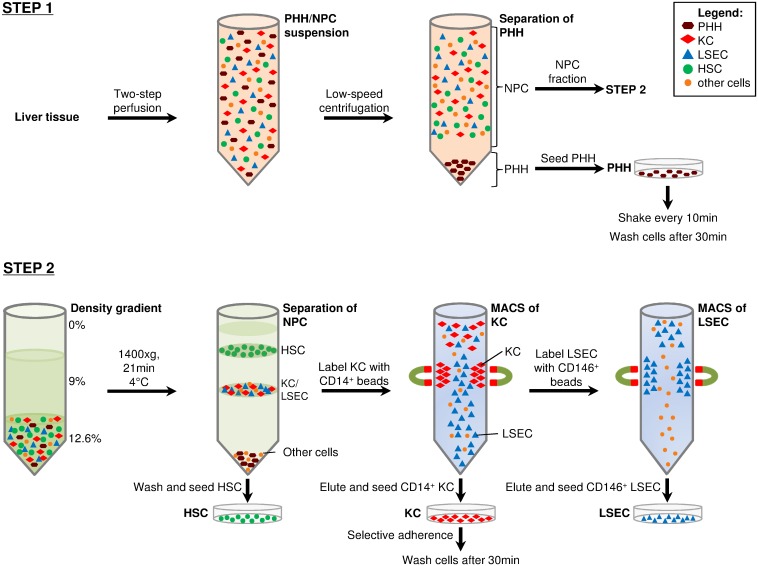
Isolating and culturing primary human hepatocytes is an essential tool to measure liver function, study metabolism and perform hepatotoxicity drug tests. When investigating liver diseases comparative studies of diferent cells within the same patient sample are essential. This approach has many drawbacks since the liver is composed of 80% parenchymal cells (hepatocytes) and 20% non-parenchymal cells, making it difficult to obtain cellular fractions representative of all the subsets present in this organ. Additionally, the obtained cellular fractions usually lack the purity and viability necessary to analyse cell-specific features.
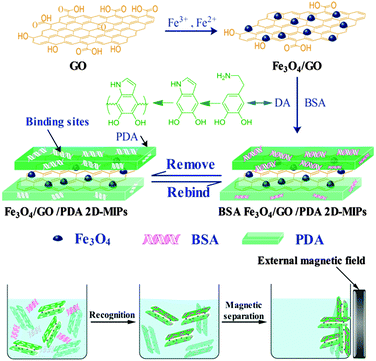
Removal of a protein from solution requires specific recognition of the protein. This has been accomplished through the use of antibodies, epitopes, and functional moieties for years. Now there is a new tool available. It is called a molecularly imprinted polymer (MIP). MIPs have cavities in the polymer matrix that are the perfect fit for the target protein. They have all the right functional groups in all the right places, and this is due to the way they are made.

At the end of the year 1959, Richard Feynman presented his talk entitled There's Plenty of Room at the Bottom to the brightest physicists of the time. The conference participants shared a general feeling that the greatest advances in science had already been made. In a sense, they felt that the good discoveries had already been taken and only scraps remained for them. Feynman challenged this attitude by suggesting that there were more discoveries waiting to be made than they could even imagine. To find them, they needed to look at the nanoscale.
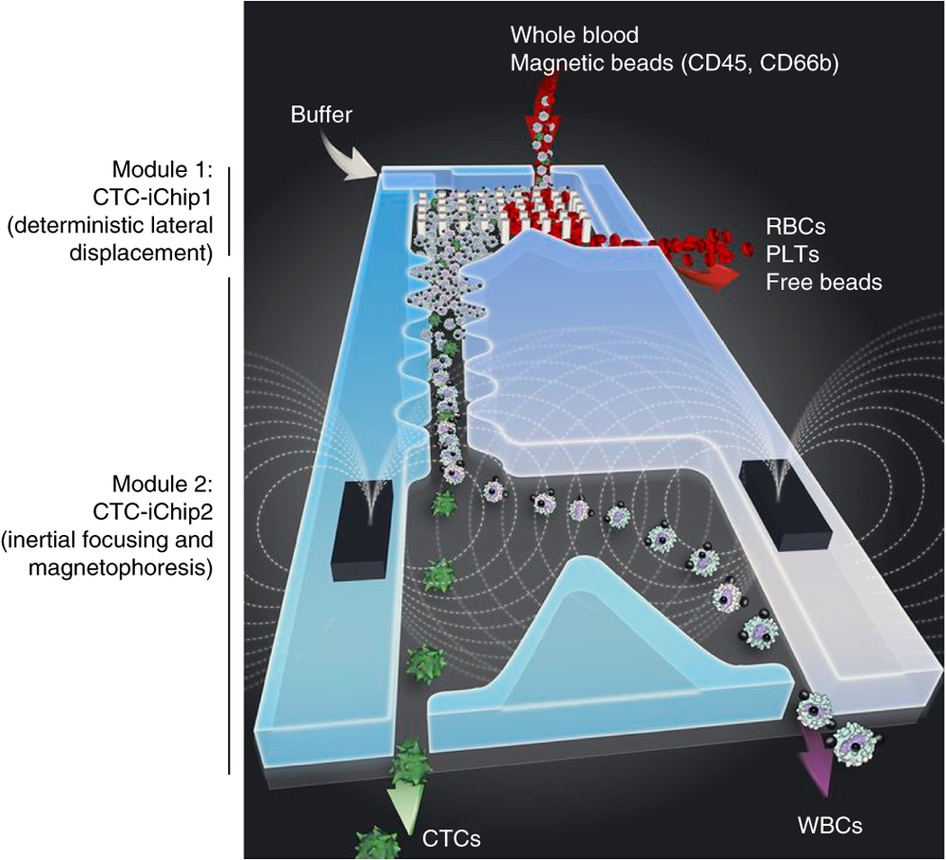
The isolation of circulating tumor cells is essential to diagnostics and metastasis studies. Since these cells are rare compared to the whole cell content of blood, the isolation process poses a challenge. There are several widespread methods of analysis and isolation of circulating tumour cells, from direct analysis of blood by marking tumor cells selectively, to blood processing through flow cytometry and magnetophoresis, that are based on EpCAM expression by tumor cells. However, circulating tumor cells from cancers of non-epithelial origin and cells that go through epithelial to mesenchymal transition do not have EpCAM. Therefore, these approaches have the tendency to lose rare cells.