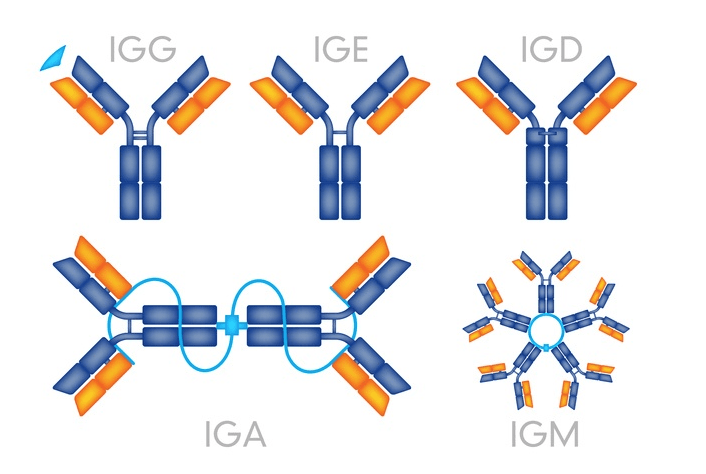
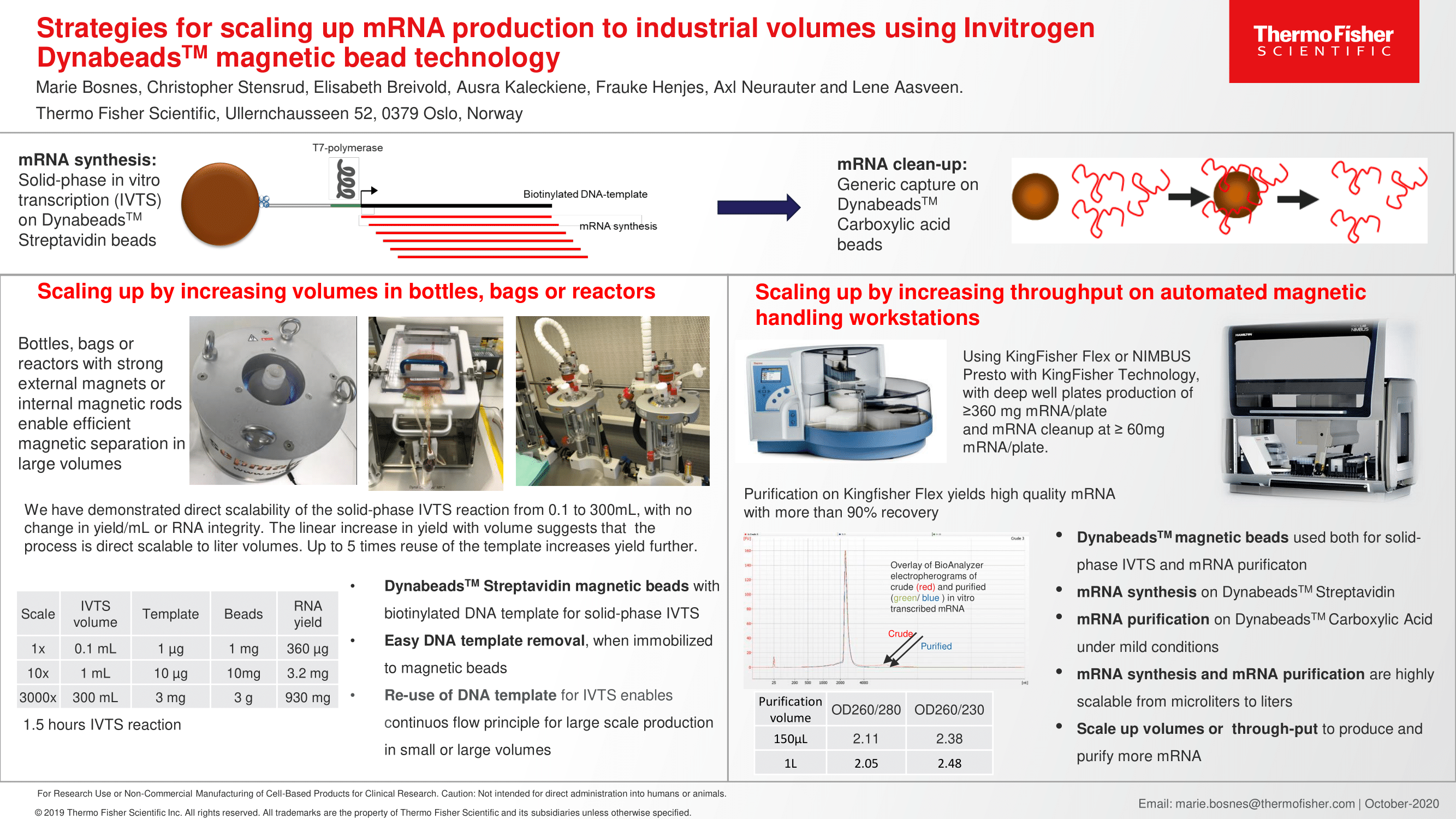
We have come a long way from the days of blood letting, trephination, and snake oil salesmen peddling cure-all tonics. The oversight and regulation of organizations such as the European Medicines Agency and the Federal Drug Administration (FDA) have significantly improved the quality and safety of our medical and pharmaceutical products. Of course, our medical understanding has deepened dramatically, our science has become more sophisticated, and we have developed tools to perform large scale drug discovery and screening. With this deeper understanding of chemistry and drug development we have realized the importance of preserving the chemical molecules via proper storage conditions.
The ICH guidelines for stability lay out the requirements for identifying and maintaining drug efficacy by understanding the pathways of degradation. The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) was founded in 1990. The European Commission, FDA from the USA, and the Ministry of Health, Labour, and Welfare (MHLW), which later became the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan are all founding members. Since that time, many other regulatory authorities from around the world have joined the ICH. The stated mission of the ICH is to “achieve greater harmonisation worldwide to ensure that safe, effective, and high quality medicines are developed and registered in the most resource-efficient manner.”