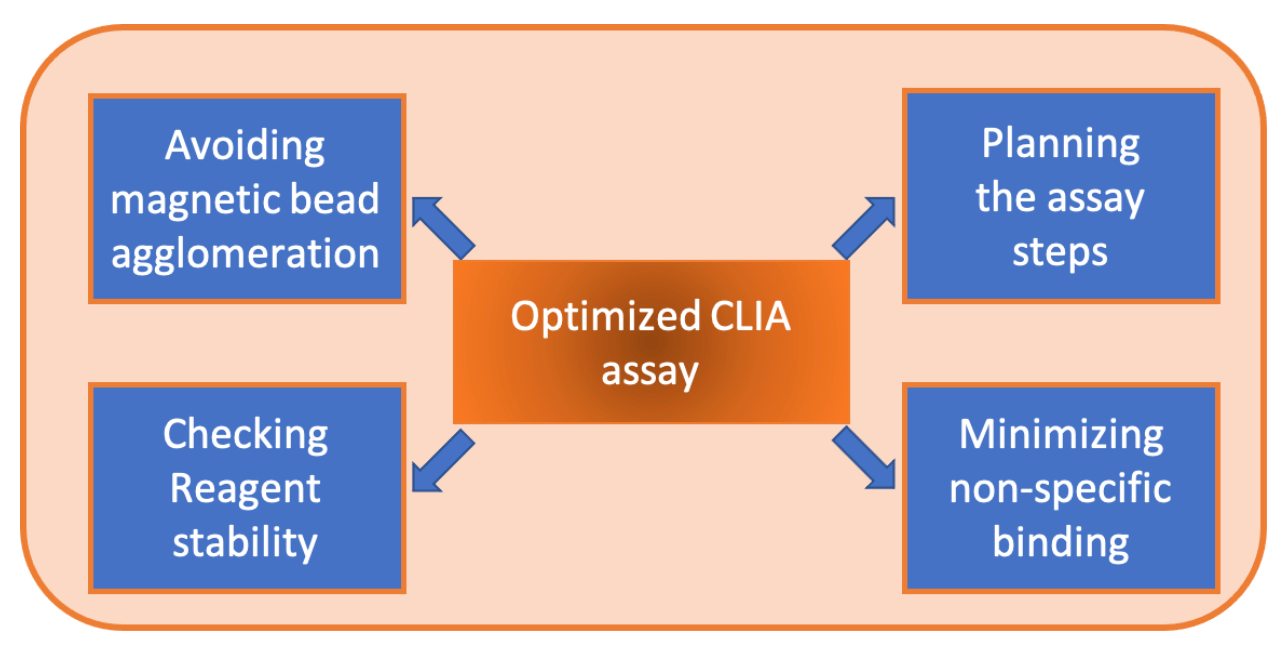
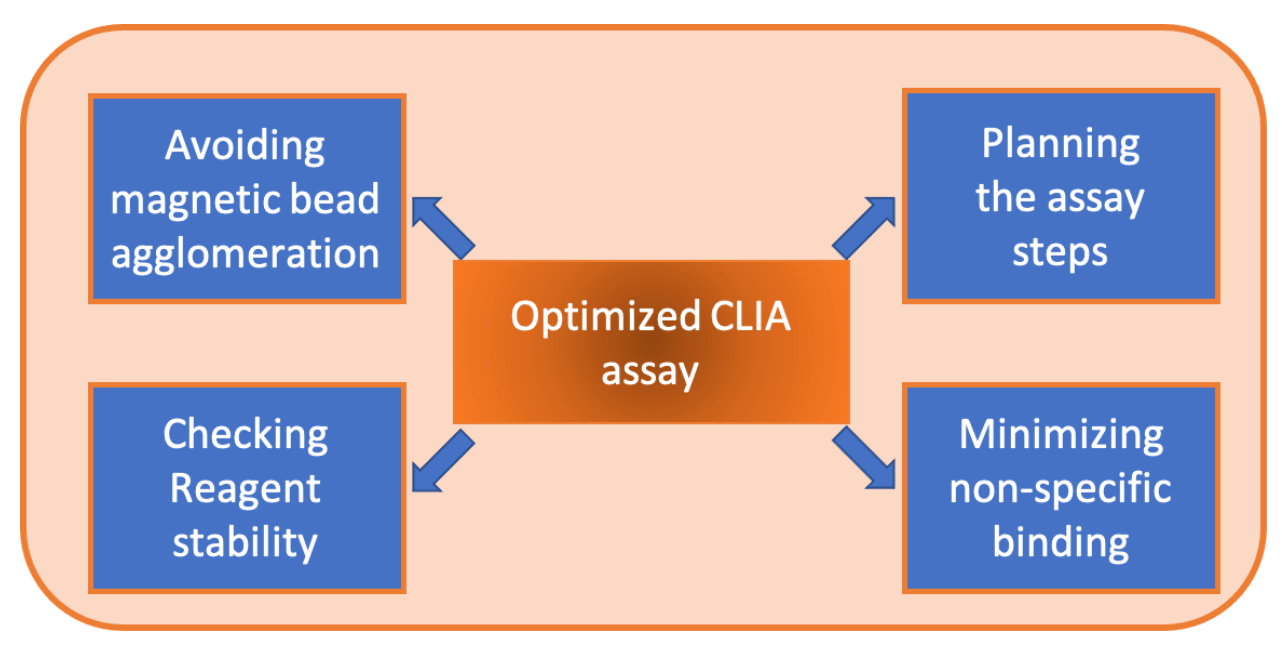
There are a few more considerations for optimizing the CLIA assay that in this chapter will be discussed. The following considerations are related to the performance of your assay.

CLIA Performance Considerations
- Proper suspension of the magnetic beads- When magnetic beads are on board the analyser and are not properly homogenized by the analyser mixer, wrong results in the assay can be obtained. This can be called the “first sample effect”, where reactivity is too high or too low on the first replicate. If magnetic beads are not properly homogenized, the quantity of particles in the assay will be different than the required. This problem needs to be troubleshooted as early as possible in the preliminary phase of the project. Many factors can modify this effect, such as particle size of the bead,the conjugation procedure or final resuspension buffer of the beads.
- Using one step or two step assay- Depending on the analyte interested to be detected, a one or two step assay may be the prefered option. A two step assay is a good option when there is concern about non-specific binding because the washing steps can help to reduce it.
- Other non-specific binding reduction methods- It is possible to include other antibodies in the assay to minimize unwanted binding events. The non-specific binding of heterophilic and human anti-mouse antibodies (HAMA) can be reduced using a mixture of polyclonal and mouse monoclonal antibodies. This mixture is usually included in the assay buffer formulation.
- Check reagent stability- As is mentioned earlier in this ebook, the stability of your assay is vitally important. It is crucial to check how the reagents can handle transport and time spent unused. To have an idea of the reagent stability, accelerated stability is performed usually exposing the reagent at high temperatures such as 37 or 46 degrees for a period of time. The decrease of reactivity under these conditions can be related to low real stability. Of course, real time stability and transport simulation need to be assessed and can not be replaced by the accelerated stability data.
In some cases it can be really challenging to obtain acceptable real time stability for the reagent and some companies try to stabilize their reagent using lyophilization.
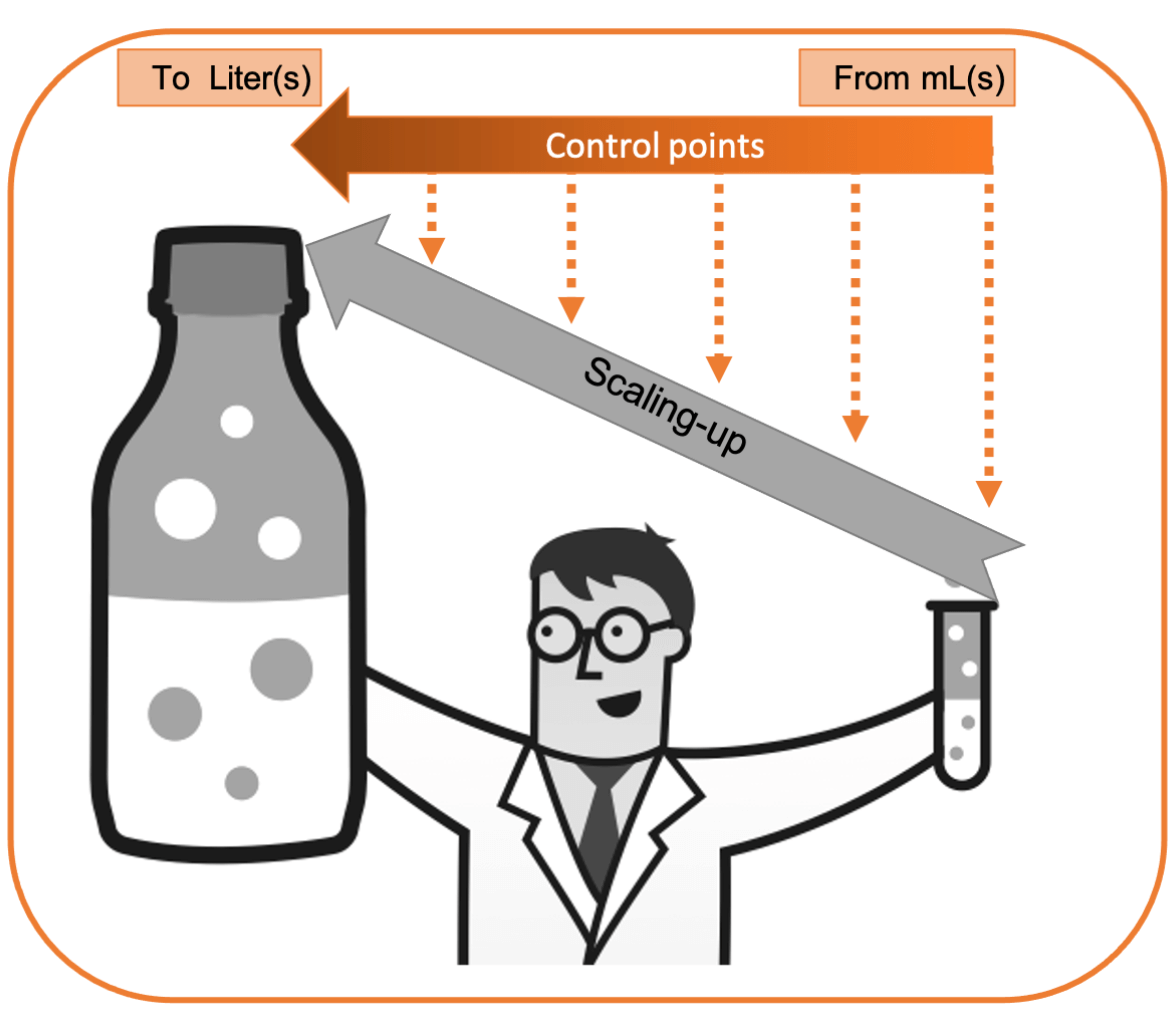
CLIA Scaling-up considerations
- Control Points- Throughout the scaling up process it is useful to have control points at which you access the variabilities in the process and in your materials. This will aid in troubleshooting when increasing the scale of each part of the process.
- Instrumentation Consistency- It is better to use similar types of instrumentation to ensure that the process will still work the same way when going from small to large batches.
- Key reagent scaling- The key reagents of the assay, such as the CLIA-labelled protein will be needed in larger quantities for scaling-up. Before scaling-up the entire procedure, it is important to be sure that the key reagents like the CLIA- labeled proteins can be successfully scaled up. .
- Other potential troubleshooting- For scaling-up, everything will need to increase. Storage capacities, temperature controlled areas, magnetic separators, and other crucial materials will need to have the capacity to be scaled-up for the whole process to move in the larger direction.
Related news
- Moving from Latex Particles to Magnetic Latex Particles
- Aspects of available Chemiluminescent immunoassay (CLIA) kits in the market
- Streptavidin HRP