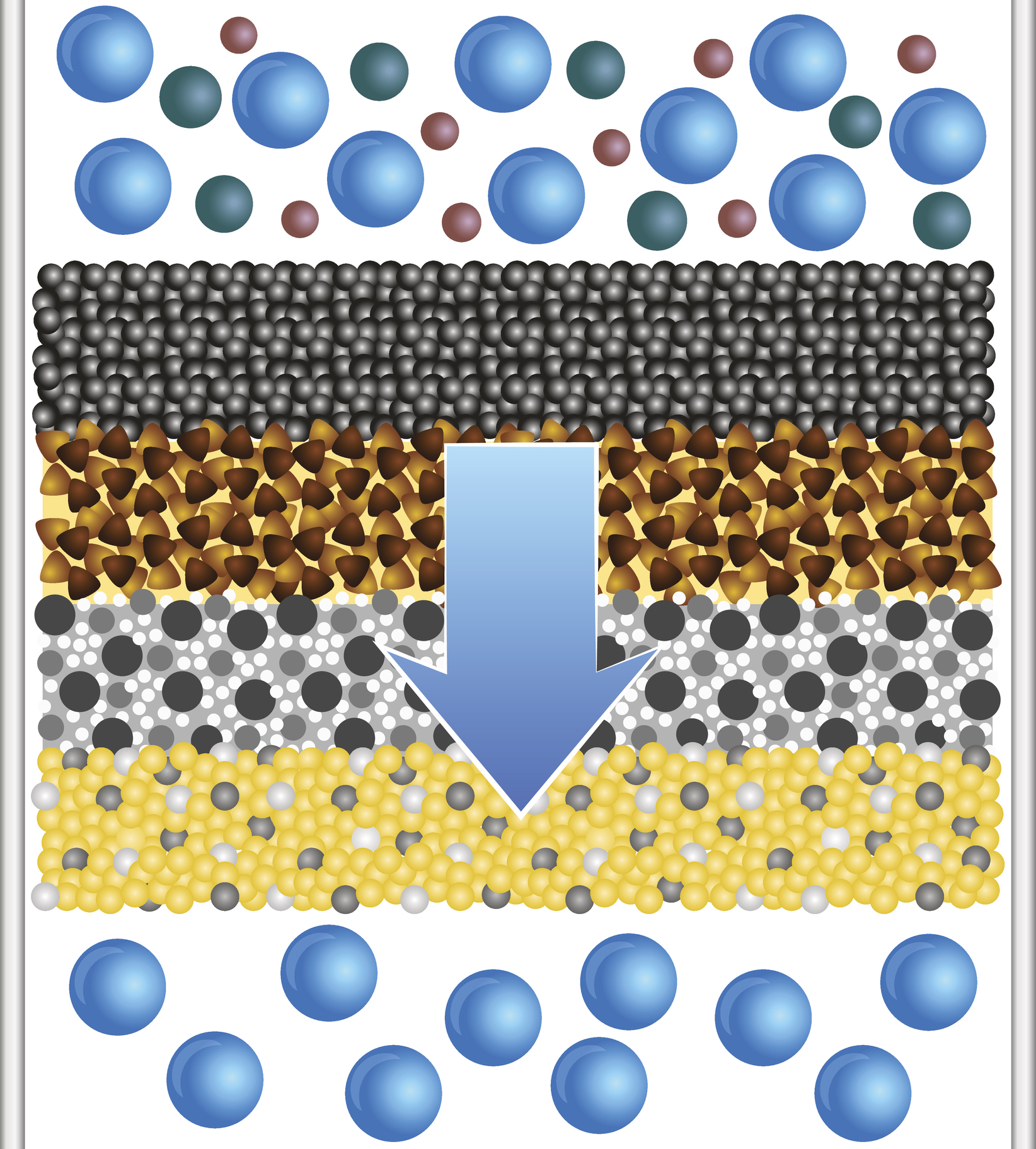
Filtration is a simple technique used to separate solid particles from suspension in a liquid solution. There are many filtration methods available, but all are based on the same general principle: a heterogenous mixture is poured over a filter membrane. The filter membrane has pores of a particular size. Particles larger than the pores will be unable to pass through the membrane, while particles smaller than the pores will pass through unhindered. Additionally, all liquids will pass through. The final result of a filtration process is a collection of residue on the filtration membrane. This residue is therefore effectively separated from the rest of the mixture that passed through the membrane.
The filtration process can be mediated by the force of gravity. This is the simplest way to achieve a separation. A common example is the filter paper used in drip coffee makers. The coffee grounds are larger than the pores of the coffee filter so they stay in place while the hot water can pick up the coffee oils, flavors, and caffeine molecules and travel through to the pot below.
Variations in the filtration process
In the laboratory it is often impractical to wait for gravity to separate a mixture. In these cases we can use a filtration process that employs a vacuum to pull the liquid and small particles through the pores of the filtration membrane. The suction of the vacuum greatly improves the speed of the filtration process. Similarly, a filtration centrifuge can be used to rapidly separate a mixture. The centripetal/centrifugal force of the centrifuge pushes the liquid and small particles through the filter membrane while the large particles remain. Some filter membranes are designed to retain the desired particles above the membrane (pore size smaller than the desired population, but larger than the contaminants), while other membranes are designed to allow the desired population through (pore size larger than the desired population, but smaller than the contaminants).
The most important step of the filtration process is determining the size of the particle you are trying to separate. Then you can choose a filter membrane with an appropriate pore size. Filter membranes can be extremely small, on the order of micrometers, which is around the size of an individual cell. The filtration process should be chosen with the goal of maintaining the viability of separated products. For example, the use of vacuum filtration is perfectly fine for chemical products, but may not be optimal for cell isolation. The size and polydispersity of submicrometer particles can be quickly measured via light scattering techniques. This will provide a general idea of the membrane size required for the filtration process. If a sample of nanoparticles is too polydisperse, meaning that the particles are all different sizes, then filtration might be an easy way to separate the nanoparticles into more monodisperse fractions. Some applications may need a more tightly controlled diameter range of nanoparticles, and a simple filtration process may simplify the overall synthesis procedure.
When it comes to cell filtration a vacumn or centrifuge may put too much force on the cells and could cause damage to the cell membrane. Biomagnetic separation could be used in lieu of a lengthy filtration process for cell separation and population enrichment. The benefits of biomagnetic separation include specificity, speed, and product viability. The superparamagnetic nanoparticles used in biomagnetic separation are easily surface-functionalized to specifically target a specific cell, substrate, or molecule. Another great benefit of biomagnetic separation is speed of separation and cell viability. It is important to note that these benefits are only seen with well-engineered separation racks that produce a homogeneous force throughout the working volume. A properly engineered biomagnetic separation rack won't cause cell lysis or bursting because the force exerted on the cells is the same throughout the working volume; cells closest to the walls of the flask will not experience a higher magnetic force than those closest to the interior.
Another variation in filtration is cold filtration. One can also call this a separation process. Cold filtration is done at cold temperatures, the solution can be cooled in an ice bath and the filtration apparatus is cooled as well. This allows small crystals to form for easy removal. For example, this technique is used for the filtration of beer. Filtering at cold temperatures allows crystals of proteins and yeast particles to be removed from the beer.
Hot filtration is also a variation of filtration. For hot filtration, the funnel or filtration apparatus is heated throughout the process, as is the sample. The goal is keep everything warm enough to keep the solution liquid, to filter out any impurities as it passes through a filter. An ideal funnel to use for this won’t contain a stem, to eliminate the chance of crystals forming anytime and slow down the process. When the sample cools down, it can crystalize again, now in a more pure state.
The optimal filtration process always depends on the experimental parameters and goals. These can vary across experimental and industrial scenarios, but the basics of the filtration process remain the same; it is important to characterize the target population and know the size of the particles being filtered. Choosing a filtration membrane with a proper pore size is essential to retaining the target while filtering out the undesired contaminants.
Uses for a general filtration process
Laboratories use filtration to make sure their solutions are as pure as possible. This can also be thought of as a sterilization since it removes unwanted particles. It can also be considered the removal of unwanted solid particles from a liquid. Many laboratory experiments and procedures require as sterile of a solution as possible. Sometimes solutions are also autoclaved, which is a process that heats the solution to a high temperature to kill any organism that might be growing inside.
Filtration makes sure that there are no contaminants in the liquid. Let’s discuss some examples of reasons to use a general filtration process. Some laboratory equipment has small tubing or fragile parts which could become clogged or ruined by particles in a buffer or water that flows through the machine. Laboratories that work with RNA are very careful to work with very sterile buffers to avoid RNase contamination.
Related news
- Upstream processing
- Challenges with your Biomagnetic Separation process?
- Immunoprecipitation with magnetic beads